Enhanced Medical Information Website | Bristol-Myers Squibb
Challenge
Our pharma client needs to provide accurate and precise search results through their Medical Information Online (MIO) website to healthcare providers and physicians who submit unsolicited requests for scientific information on off-label use of their approved drugs. Their current MIO search tool had been severely restricted in an attempt to meet FDA draft guidelines – as a result, very little useful information was returned, resulting in poor usage adoption by healthcare professionals (HCPs).
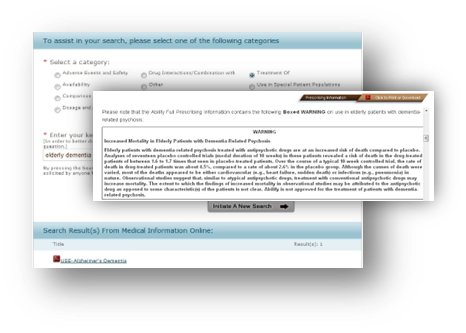
Solution
Our pharma client needed expert advice on the technical and content infrastructure that could support HCP needs, stay within FDA draft guidelines, and be extensible to multiple languages and mobile devices.
The Earley Information Science team did a series of due diligence activities before providing recommendations:
First, we reviewed the FDA draft regulatory guidelines and interpreted them from an un-biased perspective.
We conducted a competitor review to evaluate how other pharma sites presented off-label medical information. We performed a technical review of the content management and site search infrastructure, and conducted a series of interviews and working sessions to explore authoring processes and content tagging procedures.
Finally, we performed search log analysis and heuristic analysis of the MIO user experience to benchmark against search best practices for the industry.
We took our due diligence findings and created a series of short and long term recommendations for content, people, process and technology that the client could implement in their environment. Additionally, we provided technical and functional requirements that provided the criteria they needed to make technology decisions.
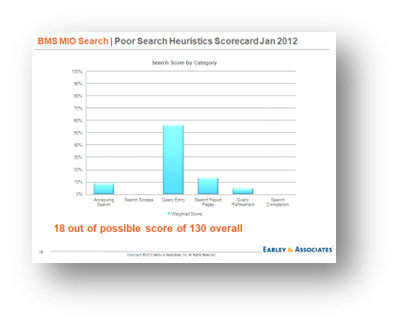
Results
Our pharma client has now embarked on a roadmap for improving the medical information search experience for health care providers. They are confident in their ability to adhere to FDA draft guidance, have a plan for exactly where to improve their technology, and have a clear understanding of the global authoring, tagging and governance processes to sustain MIO long term.

Discover Expert Insights
See the latest news in the information architecture space through our blog posts, whitepapers, webinars, and more.
